Bacteria can build slimy coatings called biofilms when they adhere to a surface. These biofilms help the bacteria resist harsh conditions and even drugs. In a recent study, scientists demonstrated how biofilm growth may be regulated by laser light in the form of optical traps. These discoveries may make it possible for researchers to use these microbial layers for a range of bioengineering purposes.
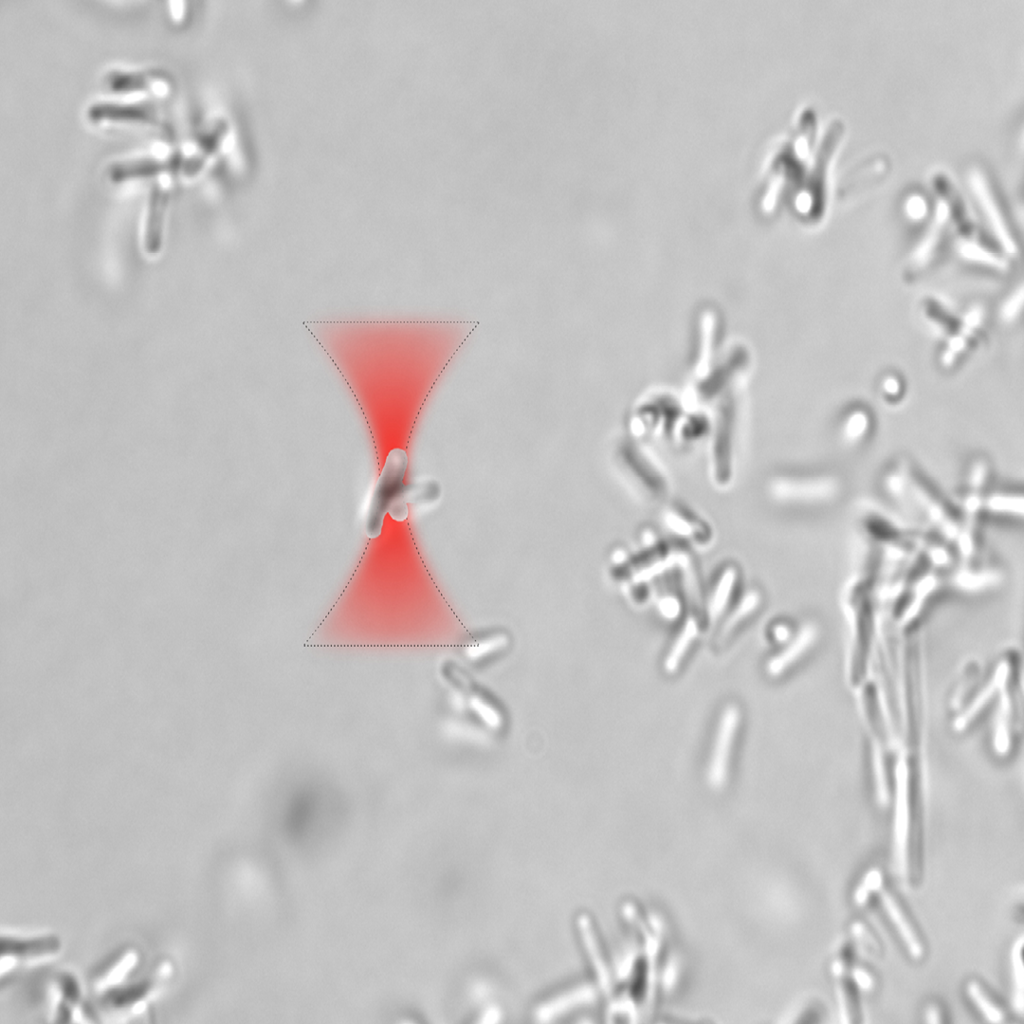
Researchers report that laser-based optical traps can be used to control biofilm formation. They found that using lasers at different wavelengths can be used to stimulate and suppress biofilm growth. Image Credit: Anna Bezryadina, California State University Northridge
Producing microscopic components usually requires a highly technical fabrication process, but we found that optical tweezers can be used to precisely control the position of individual bacteria or clusters of bacteria, and this allows us to influence the growth patterns of bacterial structures on a microscopic level with high precision.
Anna Bezryadina, Research Team Leader, California State University Northridge
The researchers describe their work on controlling bacterial aggregation and biofilm formation using optical traps in the Optica Publishing Group journal Biomedical Optics Express. They discovered that biofilm formation may be stimulated or suppressed by using various types of lasers.
We can even create a sort of bacterial Lego block that can be moved around, stuck together, and destroyed as needed, this work could lead to new types of biodegradable materials or a new generation of biofilm-based biosensors, for example.
Anna Bezryadina, Research Team Leader, California State University Northridge
Using Light to Control Bacterial Growth
Most of the research on biofilms has been on biological, chemical, and mechanical methods of suppressing and managing biofilms. Bezryadina and her team sought to know if optical methods could be used to manage biofilm dynamics, even though scientists have demonstrated that synthetic and chemical approaches may be used to activate and control biofilms and design biofilms into specific spatial configurations.
It took an interdisciplinary team with knowledge of both microbiology and cutting-edge optical technology to accomplish this.
Bacillus subtilis, a non-pathogenic bacterium that naturally produces biofilms, was the subject of the researchers' experiments. To get the bacteria to form a biofilm, they placed them in an unfavorable, low-nutrient environment that is inhospitable to B. subtilis.
They performed optical trapping studies using either a 473 nm blue laser or a near-infrared Ti:sapphire laser that was tunable between 700 and 1000 nm after producing tiny biofilm clusters.
Researchers discovered that extended optical trapping of biofilm clusters was possible with a laser emitting between 820 and 830 nm in wavelength, with the least amount of noticeable photodamage.
But when a laser of 473 nm was applied, the bacteria greatly absorbed this wavelength, which led to the rupture of the cells and the disintegration of the biofilm clusters. They also noticed that three to fifteen cells made up the optimal bacterial clusters for optical manipulation.
Making Patterns
Utilizing optical tweezers to study bacteria dynamics and biofilm formation for an hour at a wavelength of 820 nm, the researchers found that bacterial clusters accumulated close to optically captured clusters, attached to the surface, and began to form a microcolony. Additionally, they might be able to precisely relocate optically imprisoned bacterial clusters within the sample, which could help assemble bacterial structures.
The results suggest that NIR wavelengths in the 800 nm to 850 nm range could be used for optical trapping, manipulation, and pattern formation of bacterial clusters for extended periods. The NIR laser did not appear to interfere with the formation of biofilms for bacterial clusters exposed to the highly focused NIR laser.
Despite the apparent uncontrolled bacterial biofilm formation in nature, our work showed that bacterial biofilm formation can be influenced by light, and this paper represents the first step in the long-term project to create microscopic building materials from readily available resources like bacteria. In future studies, we are planning to use what we found to develop a process to construct structures from bacterial Lego blocks.
Anna Bezryadina, Research Team Leader, California State University Northridge
The trials showed some degree of flexibility in the precise growth conditions, cluster sizes, and wavelengths required to manipulate the biofilms. The technique may also apply to other kinds of biofilm-forming microbes, according to the researchers.
Journal Reference
C. Camba, et.al., (2024). Biofilm formation and manipulation with optical tweezers. Biomedical Optics Express. doi.org/10.1364/BOE.510836.