Oct 26 2016
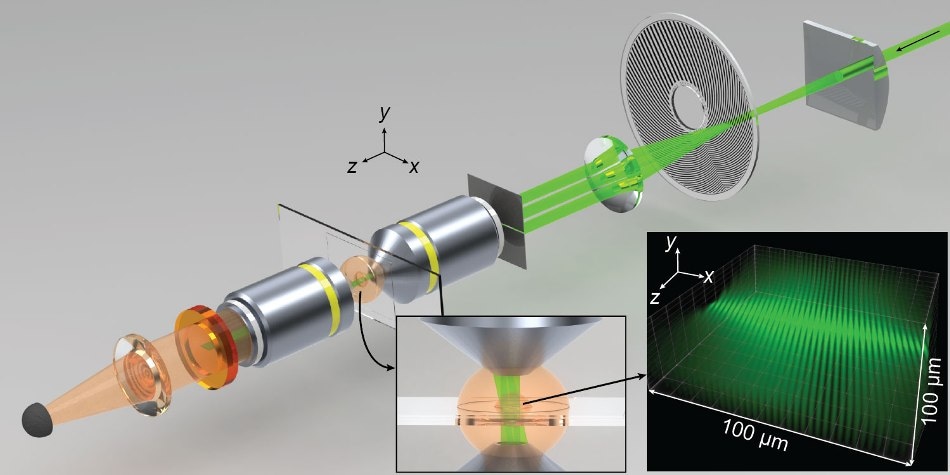 Spatiotemporal modulations of illumination intensity in the CHIRPT microscope are achieved by imaging a spinning modulation mask to the focal plane of the microscope. A spatial filter placed in the pupil plane of the objective lens allows illumination intensity to form by the interference of two beams in the object plane. The microscope and illumination intensity are shown here at a snapshot in time. Credit: Colorado State University
Spatiotemporal modulations of illumination intensity in the CHIRPT microscope are achieved by imaging a spinning modulation mask to the focal plane of the microscope. A spatial filter placed in the pupil plane of the objective lens allows illumination intensity to form by the interference of two beams in the object plane. The microscope and illumination intensity are shown here at a snapshot in time. Credit: Colorado State University
Once again, optical microscopy researchers at Colorado State University are pushing the envelope of biological imaging.
Jeffrey Field, a researcher in electrical engineering and director of the Microscope Imaging Network at CSU, has designed and developed a fluorescence-detection microscope that can combine high-resolution and three-dimensional image processing that is faster than other similar techniques.
Field, Randy Bartels, a professor of electrical and computer engineering, and David Winters, a former postdoctoral researcher, collaborated on this work, which has been published in the journal of the Optical Society of America, Optica. They called their new microscope CHIRPT: Coherent Holographic Image Reconstruction by Phase Transfer.
Imaging Tradeoffs
Field and other optics researchers work together in a world of tradeoffs. For instance, multiphoton fluorescence microscopy, an advanced deep-tissue imaging technique, uses a short, bright pulsed laser focused tight to one spot, and the intensity of fluorescence is recorded from that spot. Then, the laser moves on to the next spots in order create high-resolution 3D images.
This imaging technique provides subcellular detail, however, it is rather slow because it can illuminate only one small spot at a time.
Other techniques, such as spinning disk confocal microscopy, are relatively faster because they illuminate on multiple spots, not just a single spot, and they can simultaneously scan over a larger area. These techniques, unlike multiphoton, need to collect images with a camera. Therefore, fluorescent light produced from the specimen is unclear on the camera, causing loss in resolution and subcellular detail.
Call them greedy, but Field and collaborators want it all.
Breaking Established Boundaries
The researchers’ aim is to work around these limitations - speed, resolution and field size - to get through the established limits in light microscopy.
The new microscope, designed by Field and Bartels, builds upon an earlier published technique and allows digital re-focus of fluorescent light. It sheds light on multiple points, not just one point, by using delocalized illumination over a large area. They are using the physical principles similar to holography, where scattered light is used for building a 3D image.
Using a large illumination area, after back-end signal processing, the new microscope can identify distinct light modulation patterns of various points in the field of view. It combines the signals from all the distinct patterns to create a 3D image.
The idea is that you have a fluorophore at any point in the specimen, and the temporal structure of its fluorescence will be distinguishable from all others. So you can have this huge array of fluorophores, and just with this single-pixel detector, you can tell where every one of them is in that 2D field.
Jeffrey Field, Researcher, CSU
3D, Deep-Tissue Images
So what does the new technique allow? Three-dimensional deep-tissue images with better field-depth compared to other techniques. Field-depth, like in photography, means images in the background are in sharp focus together with the main image, and the CSU scientists can work at 600 frames per second, much faster than other techniques.
Images can also be post-processed using this new microscope so that aberrations that obscure the object of interest can be removed. It’s like being able to focus an image after it’s been taken.
The new CHIRPT microscope could help biomedical researchers to generate sharp, 3-D images of tissue or cells over a larger volume compared to standard fluorescence microscopy techniques. It could also help image multicellular processes in real time compared to a standard light microscope, which could see one cell at a time.